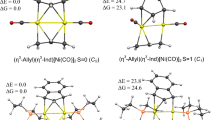
Abstract
A DFT study carried out on the diatomic ligand of InX coordinated with metal carbonyls [TM(InX)(CO)3] (TM = Ni, Pd, and Pt) using B3LYP, CAM-B3LYP, M06-2X level of theories with the basis sets 6-31G*, 6-311G**, LANL2DZ, and SDD. The NBO analysis confirms the π bonding contribution is lesser than that of σ (sigma) bonding contribution in TM–InX bond and it emphasizes the greater contribution by the indium atom. The orbital interactions occur between indium and the transition metal (TM) via σ donation TM ← InX. The WBI analysis acknowledges that the TM = InX bonds are stronger than the TM–CO bonds. The energy gap between the HOMO–LUMO is predicted by FMO analysis. The EDA analysis confirms that the ΔEelstat (electrostatic interaction) is more than that of ΔEorb (covalent bonding) of the complexes.
Graphic abstract





Similar content being viewed by others
Change history
25 August 2020
The original article can be found online.
References
Aldridge S (2006) Platinum complexes featuring terminally bound Ga+ and In+ ions. Angew Chem Int Ed 45(48):8097–8099. https://doi.org/10.1002/anie.200603643
Aldridge S, Coombs DL (2004) Transition metal boryl and borylene complexes: Substitution and abstraction chemistry. Coord Chem Rev 248:535–559. https://doi.org/10.1016/j.ccr.2003.12.003
Andrae D, Haeussermann U, Dolg M, Stoll H, Preuss H (1990) Energy-adjusted ab initio pseudopotentials for the 2nd and 3rd-row transition-elements. Theor Chem Acc 77:123–141. https://doi.org/10.1007/BF01114537
Baker RJ, Jones C (2005) The coordination chemistry and reactivity of group 13 metal(I) heterocycles. Coord Chem Rev 249:1857–1869. https://doi.org/10.1016/j.ccr.2004.12.016
Becke AD (1988) Density-functional exchange-energy approximation with correct asymptotic behavior. Phys Rev A Mol Opt Phys 38(6):3098–3100. https://doi.org/10.1103/PhysRevA.38.3098
Bergner A, Dolg M, Kuechle W, Stoll H, Preuss H (1993) Ab-initio energy-adjusted pseudopotentials for elements of groups 13–17. Mol Phys 80:1431–1441. https://doi.org/10.1080/00268979300103121
Bickelhaupt FM, Baerendst EJ, Chemie F, Marburg P (2000) Kohn–Sham density functional theory : predicting and understanding chemistry. Rev Comput Chem 15:1–86. https://doi.org/10.1002/9780470125922.ch1
Boehme C, Frenking G (1999) Theoretical studies of inorganic compounds, Part 6 = the nature of the gallium–iron bond in [Ar*GaFe(CO)4]. Chem Eur J 5(7):2184–2190. https://doi.org/10.1002/(SICI)1521-3765(19990702)5:7%3C2184:AID-CHEM2184%3E3.0.CO;2-3
Braunschweig H, Kollann C, Rais D (2006) Transition-metal complexes of boron—new insights and novel coordination modes. Angew Chem Int Ed 45(32):5254–5274. https://doi.org/10.1002/anie.200600506
Chiodo S, Russo N, Sicilia E (2006) LANL2DZ basis sets recontracted in the framework of density functional theory. J Chem Phys 125(10):104107-1–104107-8. https://doi.org/10.1063/1.2345197
Clarkson LM, Clegg W, Norman NC et al (1988) Structural studies of organomolybdenum complexes containing indium. Inorg Chem 27(15):2653–2660. https://doi.org/10.1021/ic00288a019
Cotton FA, Feng X (1998) Remarks on the gallium to iron bond in an Ar*GaFe(CO)4 molecule. Organometallics 17(2):128–130. https://doi.org/10.1021/om970971w
Dohmeier C, Loos D, Schnöckel H (1996) Aluminum(I) and gallium(I) compounds: syntheses, structures, and reactions. Angew Chem Int Ed 35(2):129–149. https://doi.org/10.1002/anie.199601291
Dolg M, Wedig U, Stoll H, Preuss H (1987) Energy-adjusted ab initio pseudopotentials for the first-row transition elements. J Chem Phys 86:866–872. https://doi.org/10.1063/1.452288
Fischer RA, Weiss J (1999) Coordination chemistry of aluminum, gallium, and indium at transition metals. Angew Chem Int Ed 38:2830–2850. https://doi.org/10.1002/(SICI)1521-3773(19991004)38:19%3C2830:AID-ANIE2830%3E3.0.CO;2-E
Frenking G, Fröhlich N (2000) The nature of the bonding in transition-metal compounds. Chem Rev 100(2):717–774. https://doi.org/10.1021/cr980401l
Frenking G, Wichmann K, Fro N et al (2003) Towards a rigorously defined quantum chemical analysis of the chemical bond in donor–acceptor complexes. Coord Chem Rev 238–239:55–82. https://doi.org/10.1016/S0010-8545(02)00285-0
Frisch MJ et al (2009) Gaussian 09, Revision A.02. Gaussian Inc, Wallingford
Gemel C, Steinke T, Cokoja M et al (2004) Transition metal chemistry of low valent group 13 organyls. Eur J Inorg Chem 21:4161–4176. https://doi.org/10.1002/ejic.200400569
Hay PJ, Wadt WR (1985) Ab initio effective core potentials for molecular calculations. Potentials for K to Au including the outermost core orbitals. J Chem Phys 82(1):299–310. https://doi.org/10.1063/1.448975
Henderson TM, Janesko BG, Scuseria GE et al (2008) Generalized gradient approximation model exchange holes for range-separated hybrids. J Chem Phys 128(19):194105-1–194105-9. https://doi.org/10.1063/1.2921797
Himmel HJ (2003) Structural motifs and reactivity of small molecules containing subvalent Group 13 elements: matrix isolation and quantum chemical studies. Dalton Trans 2003:3639–3649. https://doi.org/10.1039/B307051B
Hsieh ATT, Mays M (1971) Interaction of indium (I) halide with organometallic complexes containing metal–metal metal–halogen bonds. J Inorg Nucl Chem Lett 7(3):223–225. https://doi.org/10.1016/0020-1650(71)80194-0
Jutzi P, Burford N (1999) Structurally diverse π-cyclopentadienyl complexes of the main group elements. Chem Rev 99(4):969–990. https://doi.org/10.1021/cr941099t
Jutzi P, Neumann B, Reumann G et al (1999) Pentamethylcyclopentadienylindium (Cp*In) as terminal ligand in the chemistry of chromium. Organometallics 18(13):2550–2552. https://doi.org/10.1021/om990045c
Macdonald CLB, Cowley AH (1999) A theoretical study of free and Fe(CO)4-complexed borylenes (boranediyls) and heavier congeners: the nature of the iron-group 13 element bonding. J Am Chem Soc 121(51):12113–12126. https://doi.org/10.1021/ja992573l
Maximoff SN, Ernzerhof M, Scuseria GE (2004) Current-dependent extension of the Perdew–Burke–Ernzerh of exchange-correlation functional. J Chem Phys 120(5):2105–2109. https://doi.org/10.1063/1.1634553
Morokuma K (1971) Molecular orbital studies of hydrogen bonds III. COHO hydrogen bond in H2CO H2O and H2CO 2H2O. J Chem Phys 55(3):1236–1244. https://doi.org/10.1063/1.1676210
Pardoe JAJ, Downs AJ (2007) Development of the chemistry of indium in formal oxidation states lower than + 3. Chem Rev 107(1):2–45. https://doi.org/10.1021/cr068027+
Paularokiadoss F, Sekar A, Christopher Jeyakumar T (2020) A DFT study on structural and bonding analysis of transition-metal carbonyls with terminal haloborylene ligands [M(CO)3(BX)] (M = Ni, Pd, and Pt; X = F, Cl, Br, and I). Comput Theor Chem 1177:112750–112758. https://doi.org/10.1016/j.comptc.2020.112750
Raghavachari K, Binkley JS, Seeger R, Pople JA (1980) Self-consistent molecular orbital methods. XX. Basis set for correlated wave-functions. J Chem Phys 72:650–654. https://doi.org/10.1063/1.438955
Reed AE, Weinstock RB, Weinhold F (1985) Natural population analysis. J Chem Phys 83(2):735–746. https://doi.org/10.1063/1.449486
Reed AE, Curtiss LA, Weinhold F (1988) Intermolecular interactions from a natural bond orbital donor–acceptor viewpoint. Chem Rev 88(6):899–926. https://doi.org/10.1021/cr00088a005
Steinke T et al (2002) [Pd3(InCp*)4(µ2-InCp*)4]: three linearly arranged palladium atoms wrapped into a fluxional shell of eight InCp* ligands. Angew Chem Int Ed 41:4761–4763. https://doi.org/10.1002/anie.200290041
Su J, Li XW, Crittendon RC et al (1997) Experimental confirmation of an iron–gallium multiple bond: synthesis, structure, and bonding of a ferrogallyne. Organometallics 16(21):4511–4513. https://doi.org/10.1021/om970530c
Tuck DG (1993) The lowe oxidation states of indium. Chem Soc Rev 22:269–276. https://doi.org/10.1039/CS9932200269
Uddin J, Boehme C, Frenking G (2000) Nature of the chemical bond between a transition metal and a group-13 element: structure and bonding of transition metal complexes with terminal group-13 diyl ligands ER (E = B to Tl; R = Cp, N(SiH3)2, Ph, Me). Organometallics 19(4):571–582. https://doi.org/10.1021/om990936k
Uhl W (1993) Organoelement compounds with Al–Al, Ga–Ga, and In–In bonds. Angew Chem Int Ed 32(10):1386–1397. https://doi.org/10.1002/anie.199313861
Uhl W (2000) durch vier InR–Liganden ideal tetraedrisch koordinierten platinatom Pt [In–C(SiMe3)3]4—a Pt(CO)4 analogous compound with a platinum atom tetrahedrally coordinated by four InR ligands. Z Anorg Allg Chem 626(10):2043–2045. https://doi.org/10.1002/1521-3749(200010)626:10%3C2043:AID-ZAAC2043%3E3.0.CO;2-4
Van LE, Baerends EJ, Snijders JG et al (1994) Relativistic total energy using regular approximations. J Chem Phys 101(11):9783–9792. https://doi.org/10.1063/1.467943
Van LE, Ehlers A, Baerends E et al (1999) Geometry optimizations in the zero order regular approximation for relativistic effects. J Chem Phys 110(18):8943–8953. https://doi.org/10.1063/1.478813
Wadt WR, Hay PJ (1985) Ab initio effective core potentials for molecular calculations. Potentials for main group elements Na to Bi. J Chem Phys 82(1):284–298. https://doi.org/10.1063/1.448800
Whitmire KH (1988) The interface of main group and transition metal cluster chemistry. J Coord Chem 17(2):95–203. https://doi.org/10.1080/00958978808075858
Yanai T, Tew D, Handy N (2004) A new hybrid exchange-correlation functional using the Coulomb-attenuating method (CAM-B3LYP). Chem Phys Lett 393:51–57. https://doi.org/10.1016/j.cplett.2004.06.011
Zhao Y, Truhlar DG (2008) The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: two new functionals and systematic testing of four M06-class functionals and 12 other functionals. Theor Chem Acc 120:215–241. https://doi.org/10.1007/s00214-007-0310-x
Ziegler T, Rauk A (1977) On the calculation of bonding energies by the Hartree Fock slater method. Theor Chim Acta 46(1):1–10. https://doi.org/10.1007/BF00551648
Ziegler T, Rauk A (1979) A theoretical study of the ethylene–metal bond in complexes between Cu(1+), Ag(1+), Au(1+), Pt(0), or Pt(2+) and ethylene, based on the Hartree-Fock-slater transition-state method. Inorg Chem 18:1558–1565. https://doi.org/10.1021/ic50196a034
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original version of this article was revised: “unfortunately co-author’s name has been published incorrectly”.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Paularokiadoss, F., Alagan, S. & Christopher Jeyakumar, T. Coordination of indium monohalide with group-10 metal carbonyls [TM(CO)3(InX)]: a DFT study. Chem. Pap. 75, 311–324 (2021). https://doi.org/10.1007/s11696-020-01297-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-020-01297-w